VIDEO: Co-leads Jennifer Lane, Jamie Weaver discuss the findings, impact and more
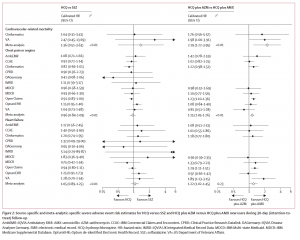
Source-specific and meta-analytic-specific severe adverse event risk estimates for HCQ versus SSZ and HCQ plus AZM versus HCQ plus AMX new users during 30-day (intention-to-treat) follow-up
The combination of hydroxychloroquine (HCQ) and azithromycin (AZM) has been linked to significant cardiovascular risks, including mortality, in the largest safety study ever performed on both HCQ and HCQ+AZM. This network study, led by the Observational Health Data Sciences and Informatics community, was recently published in Lancet Rheumatology.
In patients with rheumatoid arthritis, HCQ treatment in the short term (30 days) was found to not carry an excess risk of complications associated with its use, but HCQ treatment in the long term had a 65% relative increase in cardiovascular-related mortality, compared to sulfasalazine.
HCQ + AZM had a cardiovascular mortality risk that was more than twice (2.19) as high as the comparative treatment even in the short term based on findings from more than 320,000 users of that combination therapy. This treatment also produced a 15-20% increased rate of angina/chest pain and heart failure.
The full paper is available here.
This study, first released on MedRxiv, has already made significant impacts in the healthcare community. On April 23, the European Medicines Agency (EMA) cited the study in a warning about the risk of serious side effects with chloroquine and hydroxychloroquine. In July, the EMA again highlighted the study, among other efforts within the OHDSI community, in its eighth revision of The European Network of Centres for Pharmacoepidemiology and Pharmacovigilance (ENCePP) Guide on Methodological Standards in Pharmacoepidemiology.
This is the first published study to be generated from the OHDSI COVID Study-a-thon, a global effort in March to set the foundation for OHDSI efforts to design and execute network observational studies around characterization, patient-level prediction and population-level effect estimation to inform decision-making around the global pandemic. Multiple studies, several of which are highlighted later, have been posted to MedRxiv and are currently under peer review.
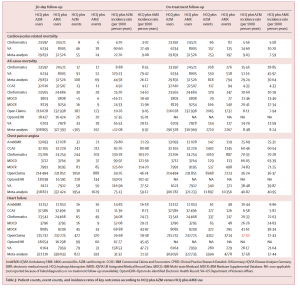
Patient counts, event counts, and incidence rates of key outcomes according to HCQ plus AZM versus HCQ plus AMX use
HCQ, a drug commonly used in the treatment of malaria, lupus and rheumatoid arthritis (RA), gained early attention during the pandemic as a potential COVID-19 treatment. The short-term (<30 days) safety profile did not identify excess risk in any of 16 severe adverse events as compared to a similar RA drug, sulfasalazine (SSZ). Long-term HCQ therapy was associated with a 65% increase in cardiovascular mortality as compared to SSZ.
“Hydroxychloroquine, both alone and in combination with azithromycin, gained strong consideration as a potential COVID treatment without a large-scale study of its overall safety profile,” said Daniel Prieto-Alhambra, MD, MSc, PhD, co-senior author on this study. “We had access to an unprecedented amount of data on this drug, and we were relieved to find no worrying side effects in the short-term use of hydroxychloroquine. However, when prescribed in combination with azithromycin, it may induce heart failure and cardiovascular mortality and we would urge caution in using the two together.”
This study examined more than 950,000 HCQ users through deidentified electronic health records and administrative claims data over a 20-year period. Records were collected from 14 different databases spanning six nations (Germany, Japan, Netherlands, Spain, United Kingdom, United States) and then mapped to the OMOP Common Data Model to generate this large-scale analysis.
“At medical school we were taught to ‘first do no harm’ and to me, our study focuses on this core belief of modern medicine,” said Jennifer Lane, MD, who served as co-lead author on this study along with Jamie Weaver, MPH, MS. “OHDSI has the power to investigate this question in a very thorough way and to go through rigorous steps. We are looking at patients from the general population, which is why it is so important to look at data from multiple countries. There are reasons why you may get bias from one data source, but if we find a signal in the Netherlands, and we find it in Spain, and we find it in the U.S., then we know we have something.”
The study was developed and executed by the OHDSI (Observational Health Data Sciences and Informatics) community, a multi-stakeholder, interdisciplinary collaborative to bring out the value of health data through large-scale analytics. All solutions are open-source, and links to the study protocol, code and results are posted at the bottom of this release.
“It required a global effort to generate this level of reproducible, reliable real-world evidence to inform decision-making around COVID treatment,” said Patrick Ryan, PhD, co-senior author on this study. “Our community collaborated for years to develop the high-level analytics which set the course for these studies. Standardizing data for nearly 1,000,000 patients on hydroxychloroquine provides confidence in these findings, and we are pleased to see that this study has already helped make a positive clinical impact as treatment options continue to be evaluated.”
More OHDSI network studies, including a multi-institutional characterization study of hospitalized COVID patients and prediction models that would assist in hospitalization decision-making, have been posted to MedRxiv and are currently under peer review. All updates on OHDSI efforts on COVID-19 will be posted here and within OHDSI community forums, as well as shared on both the community Twitter and LinkedIn feeds.
More Information
Daniel Prieto-Alhambra, MD, MSc, PhD, is Professor of Pharmaco- and Device Epidemiology at the University of Oxford.
Jennifer Lane, MD, is Orthopaedic Registrar and Versus Arthritis Clinical Research Fellow at the University of Oxford.
Jamie Weaver, MPH, MS, is Associate Director, Observational Health Data Analytics at Janssen Research and Development.
Patrick Ryan, PhD, is Vice President, Observational Health Data Analytics at Janssen Research and Development and Adjunct Assistant Professor of Biomedical Informatics at Columbia University.
All other authors can be found in the paper.
The study, “Safety of hydroxychloroquine, alone and in combination with azithromycin, in light of rapid wide-spread use for COVID-19: a multinational, network cohort and self-controlled case series study,” was published Aug. 21, in Lancet Rheumatology.
